 
Here the reagent is a constant direct electrical current of known magnitude that consumes the sample. The physiochemical phenomenon involved to attract and hold a gas, vapor, or liquid on the surface of a solid, particularly on a finely divided material. Chemical change changes caused as the result of a chemical reaction a new substance is. This involves measuring mass of reagent that reacts completely with the sample. Viscosity resistance of a liquid to shear forces (flow). 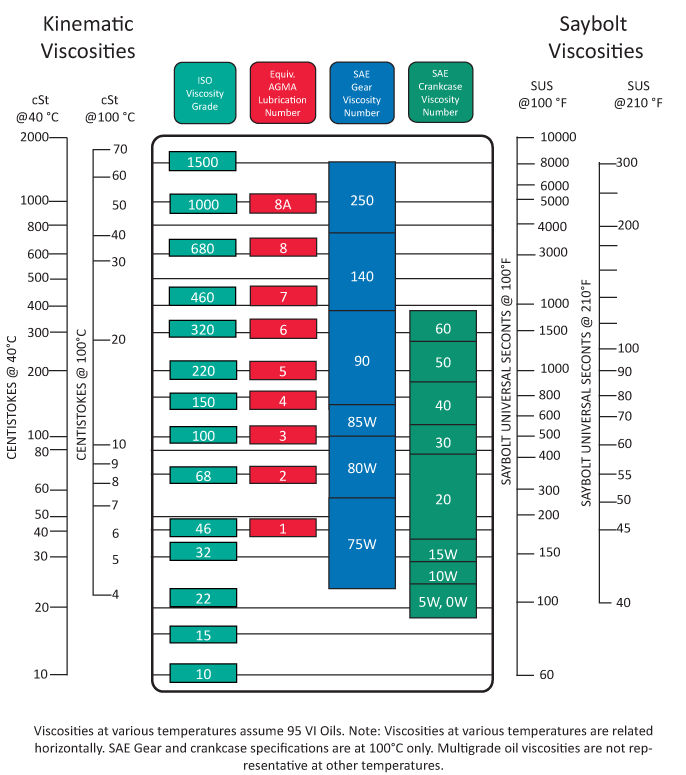
Indicators are added to the solution mixtures to produce an observable physical change at the end-point or near equivalence point ii. The two values are usually different and the difference gives the titration error. Equivalence point is a point in titration when the amount of standard solution added is exactly equal to the amount of the sample whereas end-point is the point in titration when a physical change occurs that is associated with a condition of chemical equivalence. Molasses, for example, has a greater viscosity than water. The reciprocal of the viscosity is called the fluidity, a measure of the ease of flow. Equivalence point and end-point are confused to mean the same but they are totally different. viscosity, resistance of a fluid (liquid or gas) to a change in shape, or movement of neighbouring portions relative to one another. Back titration is the process by which the excess of the standard solution used to consume the sample is determined by titration with a second standard solution. Titration is the process in which the standard reagent is added to a solution of the sample until the reaction is judged to be complete. The reagent of exactly known concentration is refered to as standard reagent. This involves measuring volume of solution of known concentration that is needed to react completely with the sample. Titrimetric methods include powerful group of quantitative procedures that are based on measuring the amount of reagent consumed by the analyte. A fluid with low viscosity flows easily because its. The volume or mass of the reagent needed to react completely with a fixed quantity of the analyte is obtained from which the amount of analyte is determined. A fluidwith large viscosity resists motion because its molecular makeup gives it a lot of internal friction. The viscosities of some representative liquids are listed in Table 11.3.1 and show a correlation between viscosity and intermolecular forces. 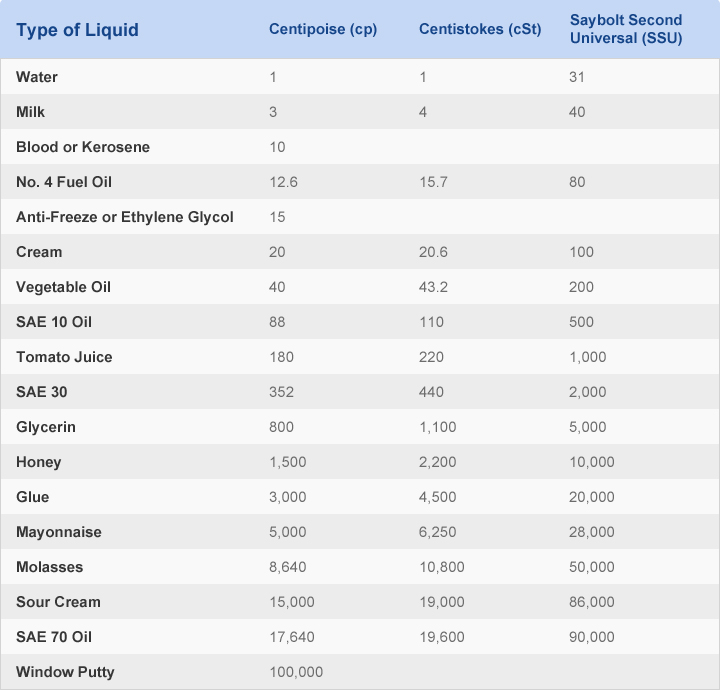
Viscosity is expressed in units of the poise (mPas) the higher the number, the higher the viscosity. Titration is based on a reaction between the analyte (unknown sample) and the regent of known concentration and reaction stoichiometry. The higher the viscosity, the slower the liquid flows through the tube and the steel balls fall. TITRIMETRIC MTHODS Titrimetric methods are widely used in chemistry to determine oxidants, reductants, acids, bases, metal ions, etc. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
